Abstract
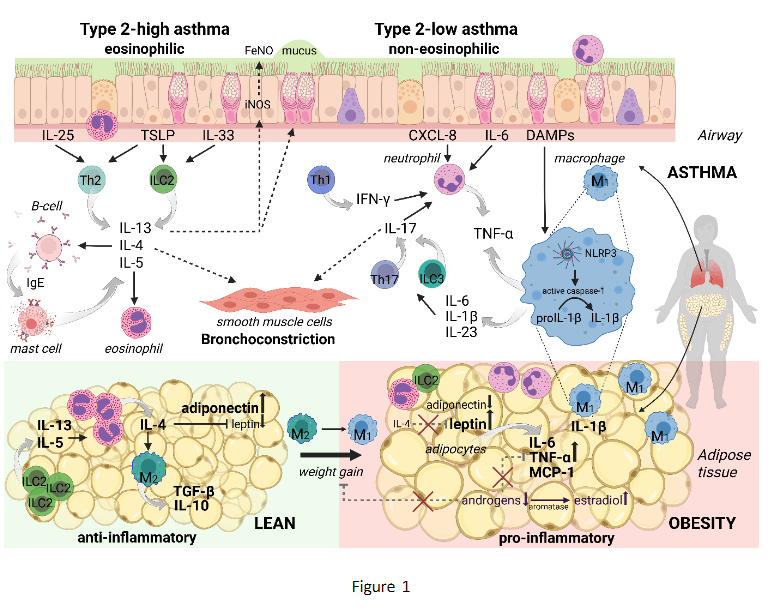
Obesity affects more than 650 million adults worldwide, with prevalence continuing to rise across all age groups and continents. This trend has important implications for asthma: individuals with obesity have a 30–50% higher risk of developing asthma, and obesity is highly prevalent among people with established disease. Mean Body Mass Index (BMI) in clinical trials and registries of adults with asthma consistently ranges from 28–30 kg·m−2, with up to 70% of patients being overweight or obese. These numbers highlight obesity as one of the most common comorbidities in asthma, consistently associated with poorer asthma control and a higher risk of exacerbations. Although obesity-associated asthma is often described as Type-2 (T2)-low phenotype, it is increasingly recognized as a heterogeneous condition not restricted to a single phenotype. Excess adiposity influences asthma through multiple mechanisms, including dysregulated adipokine signaling, impaired ILC2-eosinophil-macrophage crosstalk in adipose tissue, systemic low-grade inflammation, metabolic dysfunction, and mechanical effects on lung volumes. This diversity complicates diagnosis, endotyping, and treatment stratification. Obesity should therefore be considered a treatable trait in asthma. Weight reduction - through lifestyle interventions, pharmacotherapy, or bariatric surgery - improves symptoms, lung function, and exacerbation risk across both T2-high and T2-low asthma. Importantly, patients with obesity experience similar reductions in exacerbations with anti-T2 biologics as their lean counterparts, though improvements in symptoms and lung function are variable. Future research should prioritize randomized, placebo-controlled trials evaluating GLP-1 and dual GLP-1/GIP-agonist therapies specifically in patients with asthma and obesity, and elucidate how obesity modifies inflammatory endotypes and treatment responses.